 In molecules with more than three atoms, there are many more possible geometries. From the above chart, we can see that hydronium ion is a AX3E type molecule (A central atom, X bonded atom, E lone pair on A). However, with a triatomic molecule (three atoms), there are two possible geometries: the atoms may lie on a line, producing a linear molecule, or not, producing a bent molecule. The molecular shape of H3O+ is a trigonal pyramid and electronic geometry is tetrahedral. The molecular geometry of CBr4 is Tetrahedral. CBr4 is a nonpolar molecule because of the zero net dipole moment caused by its symmetrical structure. In BF 4, the electron pair geometry for B atom is tetrahedral. In BF 3, the electron pair geometry for B atom is plane triangular. The hybridization of CBr4 is Sp 3 and the bond angle of 109.5°. B F 3 + F B F 4 involves a change in the electron pair geometry for the underlined atom. The angles between electron domains are determined primarily by the electronic geometry (e.g., 109. It states that valence electrons will assume an electron-pair geometry that minimizes repulsions between areas of high electron density (bonds and/or lone pairs). The total valence electron is available for drawing the carbon tetrabromide ( CBr4) lewis structure is 32. An example of the complexities which arise with polyatomic molecules is molecular geometry: how are the atoms in the molecule arranged with respect to one another? In a diatomic molecule, only a single molecular geometry is possible since the two atoms must lie on a line. The Valence Shell Electron Pair Repulsion (VSEPR) theory is a simple and useful way to predict and rationalize the shapes of molecules. 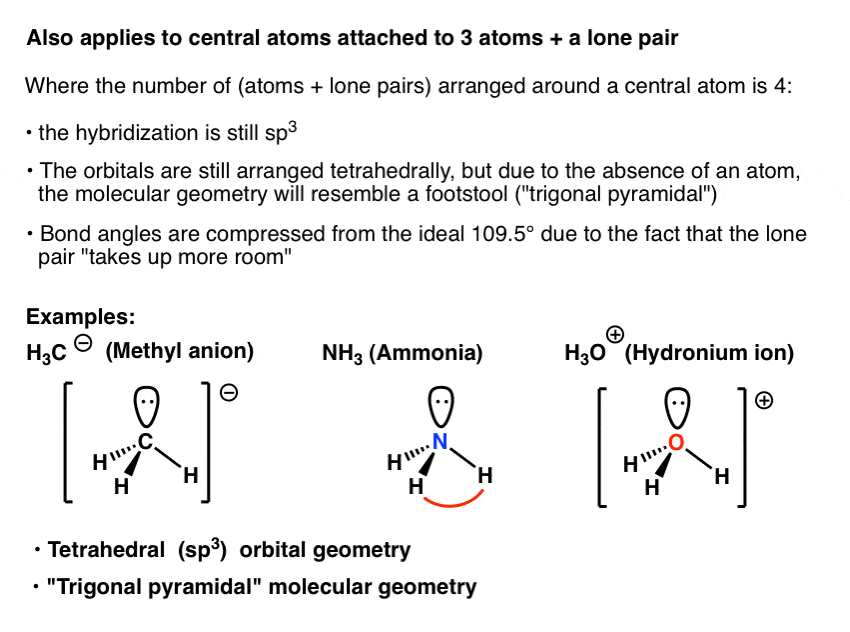
So when asked to describe the shape of a molecule we must respond with a molecular. The molecular geometry is the shape of the molecule. The electron-pair geometry provides a guide to the bond angles of between a terminal-central-terminal atom in a compound. A polyatomic molecule contains more than two atoms. Molecular geometry is the name of the geometry used to describe the shape of a molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
